A Clean Fight: The science of hygienic brewing
The most important thing a brewer does during a brew is cleaning and sanitizing. This starts before the brew and continues long after the boil has been cooled. Without chemicals we only have mechanical action to clean and the green scrubby should be a staple in any brewer’s fight against soils. Understanding cleaning processes will help us choose chemistries, clean with less labor, use less water, and certify efficacious results. Any time you start taking aggressive chemistries and using them to remove materials there is a danger involved. Understanding those dangers is extremely important. Perhaps the most dangerous sanitizing agent is hot water. Don’t be lulled into complacency.

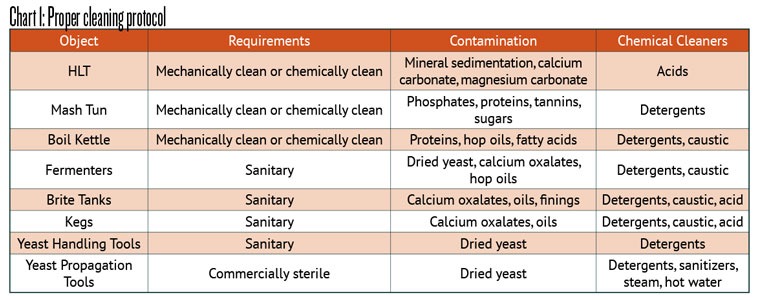
Before we can clean something it really helps to understand what it is we are removing. In the brewing process we have mineral sedimentation, mineral scale formation, protein deposits, hop resins and oils, tannins, fatty acids, dried yeasts, and more that we want to learn how to remove properly. Each part of the process has different things to remove and different requirements for cleaning. I break the levels of cleanliness into four categories: Mechanically clean (elbow grease), chemically clean (mechanically clean followed by a detergent), sanitary (chemically clean followed by a sanitizing cycle), and sterile (chemically clean followed by a sterilization step). I say sterile, but that does not exist in many food facilities. It is usually called commercially sterile and would not pass for sterile in a hospital.
Next, I find it is useful to look at each thing to be cleaned and what usually needs to be removed.
As you can see from Chart 1 below, detergents and caustics (like sodium hydroxide and potassium hydroxide) are used for the same kinds of soil. Caustics are generally faster at cleaning because they are very effective at dissolving proteins, fats, and oils, but much more dangerous than households detergents, such as dishwashing detergents. It is very important to be trained in the proper handling of caustics before use. A chemical apron, splash proof eye protection (aka goggles) and heavy-duty gloves are required. We are going to assume that most brewers reading this are using detergents and start with understanding how they are designed.
Soap is the most common detergent we have experience with but soap is not very useful to the brewer. The detergent we mostly use as a brewer (in products like PBW) is sodium metasilicate, a moderately strong alkaline cleaner, with detergent additives (aka surfactants or dispersants) to improve its properties distinguishing different brands. A detergent has a hydrophilic (water-loving) side and a hydrophobic (water-hating) side. One end of the molecule will attach to water and the other end will attach to oil and allow it to be rinsed away. We call this process emulsification. When considering a detergent we look at, at least, four properties:
- Wetting properties – How well can the solution cover a surface
- Emulsifying ability – How well can we take oils and attach them to water
- Deflocculating power – How well can we unstick things from each other
- Dissolving power – How well can we put minerals into solution
We also look for additives to help improve cleaning processes:
- Chelating/sequestering agents – Improves effectiveness in hard water by binding or chelating/sequestering calcium and/or magnesium
- Anti-foam agents – Improve the performance while pumping
- pH control – Raising the pH improves detergent action at the expense of safety
- Oxidizers – Improve the hydrophilic component of detergents. Oxidizers help dissolve protein deposits by breaking sulfhydryl groupings that help proteins cross-link; they also help remove mineral deposits from surfaces; stabilized peroxides are especially effective when combined with alkaline cleaners, like caustic
Once we take a detergent and dilute it with water it becomes a better cleaner than straight detergent or pure water. An important thing to consider is the speed that a detergent works increases with temperature up till the point that the chemistry is destroyed. Since protein soils can become “baked” onto surfaces when heated, it is a good idea to rinse protein soils with warm water before using hot chemical cleaners. A good rule of thumb is for every 10 °F (5.5 °C) you increase the temperature, you cut the cleaning time in half. In the chemistries I am familiar with the first thing to fail was the anti-foaming agent and the solution would foam if I got it too hot. Hot liquids are dangerous! I would recommend not exceeding 120 °F (49 °C) outside of a commercial setting.
Before we can clean something it really helps to understand what it is we are removing.
In a commercial setting, caustic, or caustic soda, is often used as the cleaner of choice in many applications. It works by breaking down (hydrolyzing) the proteins in soils so they can easily be rinsed away. It can also clean oils by a process known as saponification (same as in soap-making) making the oils soluble in water. There are two varieties of caustic sodas in common use — sodium hydroxide and potassium hydroxide. Bleach, or sodium hypochlorite, is oftentimes added to caustic to increase effectiveness, as bleach is an oxidizer and improves cleaning performance. Many brewers will not use sodium hypochlorite due to the low flavor threshold of chlorine compounds created by poor rinsing or wood contamination. Similar additives to detergents are often included on a brand-specific basis. It should be noted that bleach is only corrosive to stainless steel when the solution pH is acidic; chlorinated caustics do not harm stainless steel because of their high pHs. Problems often arise, however, when acid cleaning cycles follow chlorinated caustic cycles that have not been thoroughly rinsed.
Caustics rely on very high pH to clean. This makes them dangerous for eye and skin contact. The other problem with caustics is that they become ineffective when used in a CO2 environment. CO2 dissolves into the water as carbonic acid and reacts with the caustic to lower the pH. If this happens in a solution spiked with bleach the pH can go low enough so that the hypochlorite loses an oxygen molecule and is converted to chloride creating a corrosion concern.
The desire to clean in a CO2 environment (such as the interior of a keg) has led breweries to use “Acid First” cleaning methods. Acid first methods rely on mixing acid and detergent and cleaning at 1.5 to 2 pH. This is not very effective on proteins but is very useful in brite tanks and kegs. The acids used are usually phosphoric or a phosphoric/nitric blend.
After cleaning it is very important to rinse thoroughly. If you rinse insufficiently your next brew will be contaminated. At best this will cause a pH problem, at worst it will create chlorine compounds. The science of rinsing has been studied very thoroughly as it is one of the highest uses of water in the brewery. One solution is to neutralize caustic cycles with acid cycles since caustic is slow to rinse. This has the added benefit of passivating stainless at the same time. Another solution has been to measure pH of rinse cycles to verify completion.
Passivation of stainless is using an acid, usually nitric or citric, to remove free iron from the surface to promote the formation of a chromium oxide film after exposure to air. Free iron is easily oxidized to rust and a chromium oxide film helps protect stainless.
We have addressed how to remove common soils from brewing equipment but we have not talked about removing beer stone. Beer stone is the precipitation of calcium oxalate. Oxalates are present in malt and combine with calcium to form calcium oxalates. Beerstone is porous and is capable of harboring microorganisms from chemical sanitizers so its removal is more than cosmetic. Beerstone is mostly impervious to detergents as the hydrated form of calcium oxalate is completely insoluble in water. The best solution is to clean with an oxidizing acid like nitric, an oxygen producing cleaner like “Oxi-Clean,” or the old standby green scrubby.
Anything that touches the beer after boil must also be sanitized. Sanitizing is much easier than sterilizing and is sufficient for brewing beer. The goal of sanitation is to kill 99.99% of organisms not killed in contact with beer. Homebrewers have used many very simple safe sanitizers.

Sterilization is usually reserved for the most sensitive aspects of brewing, lab work, and yeast propagation. Useful tools in sterilization are an autoclave/pressure cooker with 250 °F (121 °C) steam for 30 minutes or direct flame.
If you plan to discharge cleaning chemicals to the drain it is prudent, and in some cases required, to neutralize them before discharge. Detergents are generally safe to discharge but when phosphates (e.g. TSP/90) are used as chelating agents they are bad for the environment. Caustic solutions above pH 12.5 are considered a toxic waste and should not be discharged before adjustment. Some municipalities require lower pHs — I used to adjust to below 9 with acids when I was brewing professionally. Acids are a corrosive problem and need to be adjusted above 5.0. Great care must be taken when adjusting strong acids and bases! There is potential for spontaneous boiling!
One of the advantages of understanding your cleaning chemistry is being able to save labor. It is much easier to start a pump with a proper solution to clean an object than it is to try with a scrubby. Take pride in your work, you are a brewer!


