Steeping & Soaking Grains
Steeping is the soaking of specialty malts, grains, and spices in water to extract flavors and aromas that we want to incorporate into our beer. It is, in essence, the making of a cup of tea. However, what is the best way to do this? Many different methods are used, and all have their proponents. Some recommend hot water, some recommend cold water, but there is always the question of how much time to steep; can it be steeped too short or too long?
These are several of the questions that led me to investigate steeping in a series of home experiments. I wanted to understand how time, temperature, and pH affected the extraction of flavors and aromas from these extra ingredients. I had four types of malt to work with: caramel 40 °L, caramel 120 °L, chocolate malt, and roasted barley. The caramel malts are kilned when wet such that they convert and caramelize right inside the husks. The caramel 40 °L represents the lower end of the color spectrum, and the caramel 120 °L is the upper end. The chocolate malt and roast barley is produced by kilning and roasting with a dark cocoa/coffee like flavor. It typically has a color of 350 °L.
Malt samples were prepared by grinding individual 20- gram samples of each malt to a fine powder in a typical blade coffee grinder and sealing them in paper cone coffee filters to produce a large group of identical samples in order to compare various conditions. The chocolate malt and roast barley were combined 50/50 to make a composite roast malt sample.
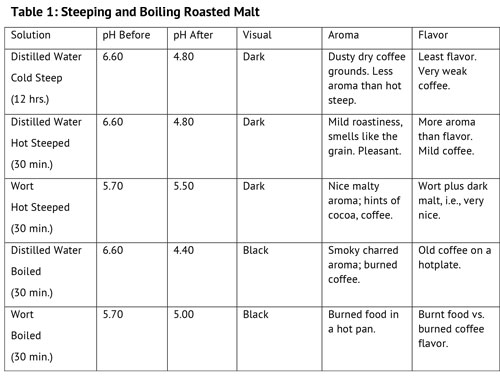
First Experiment: Steeping and Boiling Roasted Malt
For this experiment, I used the roast malt samples to investigate five extraction methods: cold water steeping, hot water steeping, hot wort steeping, boiled in water, and boiled in wort. The results are shown in Table 1. Distilled water was used for each condition to minimize external factors that could influence the perceived character of the resultant wort. The malt samples were steeped or boiled for 30 minutes because that is a typical steeping time used by homebrewers.
The results were predictable; the boiled samples were definitely the worst tasting of the group, tasting like burned coffee and burned gravy. The difference between hot and cold water steeping was similar to the difference between hot and cold steeped tea. The cold steep for 12 hours had less aroma and flavor than hot steeping, but the quality of the aroma was also different, being more like a bag of freshly ground coffee and dusty, rather than like the malt itself. The flavor of the two steepings was similar, but differed in intensity and richness of flavor. My takeaway from this experiment is that different steeping conditions will produce different flavors; that cold steeping cannot be regarded as a substitute method for hot steeping for the same quantity of malt because it tends to produce a different set of flavors and aromas. The difference in pH stability between the boiled in wort and boiled in water samples did not seem to make any real difference in the boiled roast grain flavors — they both were unpalatable. I cannot recommend the boiling of roasted specialty malts, even in decoction mashing techniques, based on results of this test.
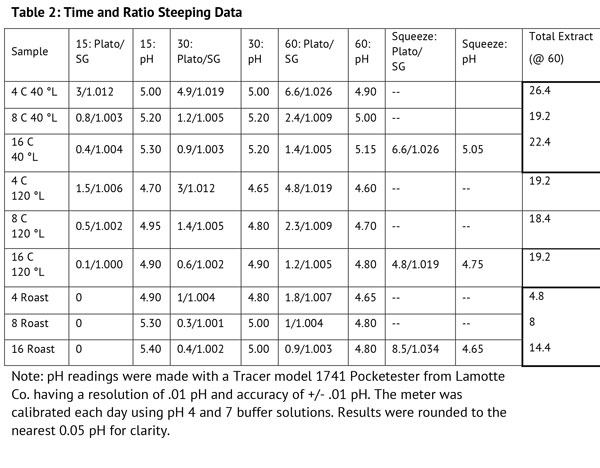
Second Experiment: Time and Concentration Effects on Steeping
The second experiment investigated the effects of water-to-grist ratio and time, using the C 40 °L, C 120 °L, and roast malt grain samples. The water to grist ratio was tested at 4, 8, and 16 liters per kilogram, which in American units is 2, 4, and 8 quarts per pound. A ratio of 2 quarts per pound is a common ratio for mashing, and 8 quarts per pound is more similar to stovetop steeping when brewing with kits. The question was, how do these variations in water to grist ratio affect the quantity and quality of the steep?
Three samples of each of the three malts were steeped in distilled water at 150 °F (65 °C) in a hot water bath at each of the ratios, and were measured by refractometer for degrees Plato and pH every at 15, 30, and 60 minutes. The data is presented in Table 2 below.
All three malt types exhibited the same general trends; that is, increasing time resulted in increasing extract. The total extract for the caramel malts were similar for each condition. The total extract for the roast malt sample on the other hand, increased with increasing ratio, indicating that concentration has more of an effect on roast malt than caramel malts.
In addition, there was a significant difference in the amount of extract that was retained in the samples (coffee filter pouches) after steeping. The sample pouches at the ratio of 16:1 L/kg were squeezed to get a sample of the retained extract, and those results were surprising. The amount of extract retained in the 16:1 samples of caramel 40 °L and 120 °L was similar or nearly equal to the amount from the same malts at a ratio of 4:1 after steeping for 60 minutes. However, the extract from the roast malt squeezing was much higher than the other measurements for roast.
The pH results for all the samples followed the same general trend, decreasing (becoming more acidic) with time. The pH of the squeezed samples did not differ greatly from the measurements at the 1-hour mark, although the drop was largest in the roast compared to the caramels. The flavor of the wort from the squeezing was not significantly different for the caramel malts but much sharper and more acrid with the roast malt.
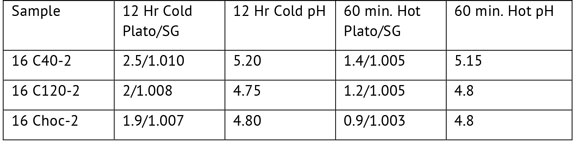
The data in this experiment were compared to samples of the same malts that were steeped cold (room temperature) for 12 hours. The extract and pH data are shown in Table 3. The amount of extract from the cold steep increased over that for the 60-minute hot steep of the malts. The pH for each malt between the two conditions was very similar. The roast malt had a lot of retained extract after the long cold steep, much more than the caramel malts.
Third Experiment: Extract and pH changes During Sparging
The malt samples were prepared as before, but instead of steeping, they were placed in the basket of a drip coffeemaker. I thoroughly cleaned the coffeemaker before the experiments and ran distilled water through several times to flush out any contamination that could affect the pH results. The malt samples were each “brewed” with 3 spargings of 320 mL of distilled water added to the coffee maker, and each sparge volume was collected in a separate pint glass for examination. The experiment was repeated for the caramel 40 °L and roast malts to see how repeatable the data was. The caramel 40 °L was almost an exact match, but the second roast malt sample was a bit different, although it followed the same trends. Even though the extract for the first sample of roast malt looks to be essentially twice as much compared to the second sample, it is really very similar considering the magnitude of the measurement. The difference in pH for the two samples of roast malt is larger, but the trend was the more important aspect of the experiment.

The caramel malts yielded the majority of their extract and color in the first sparging, with decreasing color and extract in subsequent sparging. See the photo above. The pH of both caramel malts increased, becoming less acidic, as more extract was rinsed away. The behavior of the roast malt was the opposite; the amount of extract and color actually increased with sparging, and squeezing the sample pouches afterwards showed that while the squeezed caramel malt extract potential was the same as for the last sparge, the roast malt still had significantly more extract retained in the sample. See Table 4. The third repeat was conducted a month after the first two, and the higher pH readings may be due to oxidation of the ground malt samples during that time. The samples had been stored at room temperature in a closed plastic bag.

The Chemistry of Caramel versus Roasted Malts
The color of caramel and roasted malts primarily comes from Maillard reactions (non-enzymatic browning reactions) between amino acids and sugars. These melanoidin compounds are responsible for the characteristic flavors and aromas of the malts, and are directly related to the processing temperature. The Maillard reactions produce yellow and red pigmented melanoidins at kilning temperatures less than 325 °F (163 °C), and brown melanoidins at kilning temperature greater than 350 °F (177 °C). In between there is a transition zone. It is thought that roasting temperatures (greater than 325 °F/163 °C) cause the polymerization of the yellow and red melanoidins to produce the brown, and that doing so reduces the buffering capacity, oxidation reducing capability, and acidity of the malt as the color increases. We can see evidence that would support those suppositions in the data here.
We also see evidence that indicates that the brown (roasted) melanoidin compounds are less soluble in water than the yellow/red, and likewise that the yellow/red are less easily dissolved than the malt sugars. This is evident in the visibly decreasing amount of extract with subsequent sparging in the caramel malts, and the rising amount of extract in the roast malt (see photo, above). Note that the color decreases for the caramel, but increases with sparging for the roast. This behavior was repeated in all cases, which indicates that it takes time for the brown melanoidins to dissolve and be rinsed from the roast malt sample.
Conclusions
My first conclusion is that the steeping temperature is the biggest factor for the aroma and flavor character of the steeped wort. The wort character of the roast malt changed from a dry aroma and low flavor from cold steeping, to a warm aroma, and rich flavor from hot steeping, to an oxidized and burnt character when boiled. In other words, cold steeping and hot steeping of any specialty malt will produce different flavor and aroma profiles and thus, the beers brewed using these different techniques will be different. Whether you consider one set of conditions to be better than another will depend on your personal taste as the brewer.
The steeping concentration and sparging experiments demonstrated that roast malts give up their extract more slowly than caramel malts, all else being equal. The conflicting opinions on homebrewing forums on whether the grain bag should be squeezed or not to extract all the wort may have been influenced by the types of malt they were steeping. My own taste evaluation indicates that caramel malt squeezings yield only a little more of what is already there; that the flavor of the wort doesn’t change much. Squeezing the roast malt sample though, released significantly more extract, and that extract had a lower pH and harsher flavor than the steeped wort.
The bottom line is that the brewing method makes the beer, and every change you make to the brewing method will produce a different beer. Some changes you may like, and some you may not.