Biotransformation

As a hop chemist, it breaks my heart and wounds my ego slightly to admit that when it comes to fermentation, yeast are the empresses of the kingdom. In Tarot and perhaps in a fantastical life imagined of long ago, the empress card signifies abundance, the senses, birth, fruition. The mother of all, taking in, taking care, and giving back. The yeast empress — she gives us the bounty of ethanol and, if nurtured and healthy, a wealth of secondary metabolites that exude flavor and aroma resulting in delicious, texturally balanced fermented concoctions to delight our soul. Without yeast, hops could never shine to their full potential.
These yeast, single-celled mothers bearing little buds of daughters, the future empresses of fermentation to come, possess majestic power to take one molecule and transform it into another: Biotransformation. Biotransformation, simply defined, happens when a living organism reacts or engages upon a molecule and transforms it or breaks it up into another shape. This transformation can be quite subtle as in the case of cleaving a molecule apart, or more magical resulting in the expansion or even rearrangement of the individual atoms of a molecule into a new configuration.
This word “biotransformation” is a sexy hot word in beer today – however it’s nothing new at all. It’s what yeast do to all of their substrates. It’s what our cells do to things we ingest. It’s how our body consumes alcohol and converts it into a more water- soluble form — acetaldehyde — so that we can excrete it and not die from its toxicity.
The larger kingdom of beer fermentation relies on the grand and subtle transformations biologically-driven by mother yeast (although yeast are not the sole fermenters, this article will focus on yeast fermentation). In my opinion, in beer there are four major categories of biotransformation that have received considerable attention over the past several decades. To keep the scope of this story narrow we exclude the inherent production of secondary metabolites, both good and bad. Examples of these would be diacetyl, dimethyl sulfide, and fruity esters — all of which come from raw material molecules that yeast ingest and then spit out. We will come back to esters — but from this “biotransformation” viewpoint — later.
The four categories of biotransformation most regarded of late are:
1. Glycosidic cleavage
2. Terpenol transformation
3. Organic acid conversion
4. Thiol release
Due to the dearth of literature focusing on malt and biotransformation in beer, the majority of examples outlined in this article focus on hop biotransformation by yeast.
1. Glycosidic Release of Flavor Compounds
Let’s start with what came first-ish in the literature: Glycosides. Glycosides became a hot topic about 30 years ago (although few were really talking about it) and have gained greater interest over the last two decades. The early literature that was never published in peer-reviewed journals, but rather reported in private research reports housed in the Miller Brewing Company Technical Center basement in Milwaukee, Wisconsin, is obviously hard to get access to. However, most of what could be found there was fortunately published in patents. In 1998, Miller Brewing Company’s US Patent No 5,783,235 outlined a method of “Preparing a Full Hop Flavored Beverage of Low Bitterness.”1 The secret was glycosidic release of flavor compounds via yeast treatment of spent hop material. This spent hop material is the vegetative material left over from CO2 hop oil extraction. The authors Ting, et al. went on to publish several further patents and eventually reported their findings at conferences. Their work represents some of the first literature that details the use of yeast biotransformation on hops to add hop flavor to beer, light beer specifically.
Figure 1: Structure of a glycoside

Glycosides represent a broad class of molecules considered to be flavor pre-cursors or to have flavor potential. These glycosides are water-soluble and non-volatile due to the presence of a sugar moiety (usually β–D-glucose). The organic moiety, representative of the majority of compounds found in the hop oil hydrocarbon fraction, is most often attached to the sugar moiety at its number one carbon (C-1) or glycosidic carbon (see Figure 1). Glycosides can be hydrolyzed (cleaved) to produce free aglycones (typically organic alcohols or terpene alcohols) using acid and/or enzymes. The mother yeast can also hydrolyze glycosides by excreting enzymes into their environment or onto a substrate, resulting in the release of flavor. Because the glycosides are soluble in water, they are soluble in wort and survive the kettle boil process where yeast reveal their magic. Early work done at Miller Brewing reported that hop vegetative material is made up of between 20–25% flavor-glycosides that contribute to “moderate kettle hop flavor, soft bitterness, and improved drinkability” and that the majority of hop glycosides fall into three categories: Simple organic alcohols, terpene alcohols and carbonyl compounds.2 An abundance of sensorially important glycosides of monoterpene alcohols (linalool, terpineol, and geraniol, etc.) in hops have since been identified. Having an understanding of a hop glycosidic potential is of interest as it can help a brewer better understand flavor potential from the presence of active yeast during fermentation and dry hopping.
Figure 2: Examples of glycosidically-bound aroma compounds

Unfortunately, when it comes to the majesty and biotransformative abilities, not all yeast are equal. Researchers in Belgium3,4 screened Saccharomyces and Brettanomyces brewing yeasts for hydrolase activity towards glycosidically bound volatile compounds. While only a few Saccharomyces strains demonstrated an ability to release glycosides, non-Saccharomyces strains such as Brettanomyces custersii (isolated from Lambic) possess high exo-β-glucanase activity. The authors published a list of impact aglycones from their 2008 publication — demonstrated in Figure 2. Notice the top 6 molecules, these monoterpene alcohols will be discussed in detail later. Follow-up studies have focused on glycosidic release from specific hop cultivars. Researchers from Oregon State University specifically looked at high impact American hop cultivars: Citra®, Simcoe®, Centennial, and Cascade.5 Researchers from Belgium have also looked at specific German and Japanese varieties.6 Glycosidic hydrolysis could be more active at the end of fermentation when yeasts are under nutritional stress and glycosides that make it into beer could enzymatically hydrolyze in your mouth. While there is still more work to be done, what the literature concludes today is that the addition of hop cones, pellets, and the vegetative fraction to wort and beer can lead to an increase in available hoppy flavor. The impact of this potential is largely up to the hop variety and ultimately the majesty of the yeast — yeast type, enzymatic capability, yeast health, and yeast-hop contact time.
2. Transformation of Terpenols and Terpenoid Esters
There was a time when I would give talks and the mere word “terpenes” would lose more than half the crowd. Only the hardcore hopheads would get excited about terpenoids and my nerdy hop talks. Thanks to the recent growth of the CBD and marijuana markets, and the high impact of their terpenes on flavor — it seems the word no longer aches too many heads. Terpenes are a large and diverse class of organic compounds produced by plants, and are often characterized by highly intense aroma. Hop essential oil is rich in terpenes and, as we just discussed, they also exist in the spent hop or vegetative portion of hops as glycosides. In the hop essential oil fraction, terpenes exist either in their free form or as derivatives of glucose (OK, glycosides), and/or are conjugated with other molecules such as amino acids. Some of the earliest work by such industry greats as Val Peacock indicated that terpenols such as linalool were largely responsible for hoppy flavor in beer. Hop varieties rich in linalool would thus be recommended for imparting hoppy character to beer. The issue, however, lies in the fact that linalool in its free form is volatile and blows off in the kettle. This is where biotransformation comes in to save the day.
Besides linalool there are a handful of monoterpenes, or terpenols to be specific, that are impact compounds for hoppy flavor: Geraniol, citronellol, nerol, and alpha-terpineol. As early as 1986 Lam et al. suggested that linalool, geraniol, and citronellol contribute to floral/citrus beer aroma from late hop additions. There are other terpenes such as myrcene and caryophyllene that denote strong hoppy character, but the terpenols listed here have a special connection to each other through a biotransformative pathway through interaction with S. cerevisiae.7 An illustration of this can be seen in Figure 3.
Figure 3: Biotransformation pathway of monoterpene alcohols by yeast
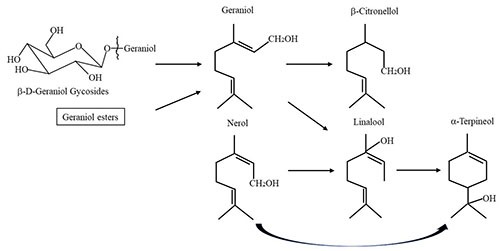
In this pathway, geraniol reveals itself as a very important intermediary terpenol, as it can be converted into β-citronelloll (citrus), as well as linalool (lavender), and nerol (lemongrass). β-citronellol is not commonly found in hops, and is typically absent in wort, however levels are known to increase during fermentation. The work of Takoi et al.8 indicates that both free and bound geraniol (enzymatically cleaved by yeast) are responsible for increases in β-citronellol, linalool, and nerol, and subsequently alpha-terpineol. As these compounds increase, they synergize to reveal hoppy citrus and floral flavor. Both lager and ale yeasts are capable of biotransforming terpenols and there is some evidence that parts of the pathway are reversible.
Brewers looking for this hoppy citrus flavor can find it in geraniol-rich hops. Geraniol is more cultivar-specific than linalool, and we still do not have a full comprehension of how much glycosidically bound geraniol exists in hop vegetative matter. North American varietals and North American-grown European varietals tend to be high in geraniol (Mosaic®, Bravo, Chinook, Citra®, Amarillo® are some prime examples). European varieties such as Saaz tend to be lower in free geraniol.9 German varieties like Comet, Hallertau Blanc, Polaris, as well as US Summit, show high potential for glycosidically bound geraniol.
We all accept and seem to understand that yeast is responsible for creation of fruity and floral esters during fermentation. While the predominant pathway for ester production relies on amino acid metabolism (think isoamyl acetate — banana), yeast secondarily metabolize free aglycones into acetate esters such as linalool acetate, citronellyl acetate, and geranyl acetate. The majestic mother yeast can further hydrolyze these as well as *ethyl esters into their sub units — acids and terpene alcohols. Yeast can hydrolyze geranyl esters into geraniol during fermentation,10 and Cascade tends to be high in geranyl esters made available only by her majesty’s biotransformative nature.
This brings us to our third topic – yeast activity on organic and fatty acids.
(*For reference: Ethyl esters are made up of short chain alcohol groups (ethanol) and a longer acid group – acetate esters are made up of a short chain acid group and a longer chain alcohol group.)
3. Biotransformation of Organic Acids to Esters to Enhance Beer Flavor
I’ve had conversations with just a handful of people regarding the impact of hop-derived branched chain fatty acids on fruity beer flavor. Most of the conversations come from anecdotal tales of brewing with old hops to achieve extremely fruity and estery flavors. This combined with Brettanomyces is one trademark of Belgian sours/lambics. I’ve always been intrigued by the floral nature Orval takes on as it ages (oxidizes). The flavors could be yeast-derived from biotransformation or also be achieved through aging as ethanol acts on organic acids to esterify them. The early patents on spent hop material may not speak directly to branched chain fatty acids, however Dr. Ting always had it at the back of his mind that they were at play and important for deriving some of the fruity estery notes in beer. My interest was peaked by an article from Zymurgy decades ago, but not many researchers seemed to have strong interest in the topic. What we know is that branched chain fatty acids originate from malt and hops and are also produced from amino acids during typical fermentation with yeast. During hop storage branched chain fatty acids increase as hops age due to the breakdown of the hop acid side chains into isovaleric acid (cheesy), isobutyric acid (vomit), and 2-methylbutyric acid (rotting fruit). During fermentation yeast convert these acids into ester form to produce fruity estery compounds: Ethyl isobutyrate, ethyl isovalerate, and ethyl 2-methylbutyrate. Ethyl esters from hop short chain fatty acids (ethyl 3 methylbutanoate, ethyl 4 –methylpentanoate) have been associated with citrus character imparted by Saaz hops.11
Recently, Takoi, beer rock star of Japan, published an article9 focusing on hop-derived fatty acids and their impact on ester formation in beer. While results of this work indicate that isovaleric acid seems more impactful on ester formation than the other acids, he and his colleagues have made an attempt at outlining a biotransformation pathway of branch chain esters by brewing yeast. An intriguing side note is that their work also revealed the impact that low levels of these fatty acids have on enhancing the flavor impact of a mixture of our terpenol friends: Linalool, geraniol, and β-citronellol. While more research is needed to fully elaborate on this pathway, it appears that evidence both scientifically and anecdotally exists to support the idea that organic acids esterification as well as ester degradation by yeast leads to increased fruity, hoppy flavor in beer.
4. Release of Bond Thiols
Although the mechanism for release of thiols is not different than the release of other bound flavor cursors, in this article thiols get their own section because of their enigma. They are also the most recent hot hop topic, really not gaining much momentum in research until about 2006 when a Belgian research group started digging deeper.12 Thiol is a chemistry term that tells us a molecule has sulfur in it, specifically a sulfur bound to an H — with the structure R-SH. Sulfur is prevalent during all fermentations, and while lots of sulfur compounds are well known in beer — dimethyl sulfide (corn), mercaptan (garbage), H2S (struck match/natural gas) — the polyfunctional thiols have other chemical moieties bound to them such as an alkyl acetate, alcohol, or other carbonyl. They are something of a different beast; they are “exotic.” These types of thiols are extremely potent even at miniscule concentrations, order of the low ppt (ng/L). The hunt for citrus-type flavor origins in hops led researchers to these thiols. Prevalent in wine grapes, thiols are responsible for aromas such as catty, foxy, boxwood, Sauvignon Blanc, mango, papaya, citrus, and other tropical notes. American or New World as well as New Zealand hops display these sought-after citrusy-type flavors in higher quantities and breeding efforts across the globe have their eyes on the thiol prize. The challenge in measuring compounds that are present at such low levels means that not many researchers have been successful in quantifying them and we are quite far away from knowing what we want to know. These thiols can be pleasant or non-pleasant, and how pleasant they are relates to chain length and structure. Roughly when the thiol is 3–4 carbons in length it tends to be less pleasant with notes of grilled meat, broth, onion, plastic, or mushroom. When the thiol has 5–8 carbons it tends to demonstrate more exotic catty, passion fruit, lemon, grapefruit, and citrusy flavor.12,13,14
A few thiols were identified early as being part of the American citrusy hop note: 3-mercaptohexan-1-ol (3 MH, citrus), 4-mercapto-4-methylpentan-2-one (4MMP, black currant or catty), 3 mercaptohexylacetate (3MHA, passion fruit)15, and 3-sulfanyl-4-methylpantan-1-ol (grapefruit)16. Gros et al.13 found 41 polyfunctional thiols among five hop cultivars: Tomahawk®, Nelson Sauvin, Nugget, Cascade, and Saaz. Each variety displayed different impact thiols at varying quantities. Tomahawk® has citrus, floral, grapefruit and black currant thiols, while Nugget seems limited to lemon, and Cascade and Nelson Sauvin meet somewhere in the middle. The authors were also the first to report finding an S-Cysteine conjugate thiol, a thiol bound to another moiety.
Today we know that thiols exist in both free and bound forms (to cysteine and gluatathione), and that the amount of thiols resulting in beer depends on whether yeast have their way with hops to release more bound forms in fermentation and dry hopping. Sorachi Ace hops, for example, are high in cysteine bound 3-suflnaylhexan-1-ol, but not so high in the free form. What this means for the brewer is that to get more grapefruit/rhubarb/passion fruit from a hop like Sorachi Ace, it needs to do a dance with yeast. To increase the citrus/lemon and grapefruit notes in Citra®, its conjugates of glutathione must be released. The German variety Polaris has large grapefruit, passion fruit, and rhubarb unleashed potential as well. While there are still so many unknowns, thiols vary in concentration by hop variety and by growing region and climate, and are sensitive to agricultural management practices (such as the application of pesticides that contain copper).
Conclusion
I always wanted to be a Hop Queen, but now knowing the potential that yeast have to release and reveal so much potential flavor from hops, I wonder if I should strive to be Empress instead? Mother yeast, nurturing, coaxing, and cleaving her way through the many interactions with molecules, biotransforming her way to delicious and alluring results. It’s worth restating — without yeast, hops could never shine to their
full potential.
References
1 Ting, P., Wilkinson, H. A., Ryder, D. S., Tripp, M. L., Rader, S., Goldstein, H. Method of Preparing a Full Hop Flavored Beverage of Low Bitterness. U. S. Patent No. 5,783,235; July 21, 1998.
2 Goldstein, H. Ting, P. Navarro, A., Ryder, D. Water-soluble Hop Flavor Precursors and Their Roles in Beer Flavor, Proc. Congress of the European Brewery Convention 1999, 27, 53-62
3 Daenen, L, 2008. Glycoside hydrolase activity of Saccharomyces and Brettanomyces yeasts. Flavor potential on hop and sour cheery glycosides. PhD Study. Catholic University Leuven, Belgium.
4 Daenen, L. Saison, D, DeSchutter, D.P., DeCooman, Verstrepen, K.J. Delvaus, F.R. Derdelinckx, G., Verachtert, H., 2008. Bioflavoring of beer through fermentation, refermentation and plant parts addition In: Preedy, V.R. (Ed.), Beer in Health and Disease Prevention. Elsevier Amsterdam, pp. 33-48
5 Daniel C. Sharp Factors that Influence the Aroma and Monoterpene Alcohol Profile of Hopped Beer, Dissertation, Oregon State University, 2017
6 Praet, T, Van Opstaele, F., Jaskula-Goiris, B., Aerts, G., De Cooman, L. Biotransformations of hop-derived aroma compounds by Saccharomyces cerevisiae upon fermentation. Cerevisia 36 (2012) 125-132.
7 King, A.J., Dickinson, J.R., Biotransformation of monoterpene alcohols by Saccharomyces cerevisiae, Torulaspora delbrueckii and Kluyveromyces lactis. (2000) Yeast 16, 499-506.
8 Takoi, K., Koie, K., Itoga, Y., Katayama, Y., Shimase, M., Nakayama, Y. and Watari, J. Biotransformation of Hop-Derived Monoterpene Alcohols by Lager Yeast and Their Contribution to the Flavor of Hopped Beer. Journal of Agricultural and Food Chemistry. 2010, 50, 5050-5058
9 Takoi, K. Behaviour of hop-derived branched-chain fatty acids during fermentation and their sensory effect on hopped beer flavours. Brewing Science Nov/Dec. 2019 Vo. 72 p. 196
10 Peacock, V.E. and Deinzer, M.L. Chemistry of hop aroma in beer. (1981) Journal of the American Society of Brewing Chemists. 34 (4), 139-141 1981
11 Kishimoto, T.; Wanikawa, A.; Kono, K.; Shibata, K. Comparison of the odor-active compounds in unhopped beer and beers hopped with different hop varieties. Journal of Agricultural and Food Chemistry. 2006, 54, 8855−8861.
12 Vermeulen, C., Lejeune, I., Tran, TT., Collin, S. Occurrence of polyfunctional thiols in fresh lager beers. (2006) Journal of Agricultural and Food Chemistry. Jul 12;54 (14): 5061-8.
13 Gros, J.; Nizet, S.; Collin, S. Occurrence of odorant polyfunctional thiols in the super alpha Tomahawk hop cultivar.
Comparison with the thiol-rich Nelson Sauvin bitter variety. Journal of Agricultural and Food Chemistry. 2011, 59, 8853−8865.
14 Tran, Hang & Cibaka, Marie-Lucie & Collin, Sonia. (2015). Polyfunctional Thiols in Fresh and Aged Belgian Special Beers: Fate of Hop S-Cysteine Conjugates. Journal of the American Society of Brewing Chemists. 73. 61-70.
15 Kishimoto, T.; Morimoto, M.; Kobayashi, M.; Yako, N.;Wanikawa, A. Behaviors of 3-mercaptohexan-1-ol and 3-mercaptohexylacetate during brewing processes. Journal of the American Society of Brewing Chemists. 2008, 3,192−196.
16 Takoi, K.; Degueil, M.; Shinkaruk, S.; Thibon, C.; Maeda, K.;Ito, K.; Bennetau, B.; Dubourdieu, D.; Tominaga, T. Identification and characteristics of new volatile thiols derived from the hop (Humuluslupulus L.) cultivar Nelson Sauvin. Journal of Agricultural and Food Chemistry. 2009, 57, 2493−2502.


