Everything About Water for Extract-Based Brewing
The best beer comes from the best water — Pilsen’s very soft water, London’s hard water, Rocky Mountain spring water, Michigan well water, Iowa spring runoff . . . or, maybe it doesn’t. Maybe the best beer comes from making the best use of the water you have. How should you treat your brewing water? Boil it first? Filter it? Buy bottled water? How much water should you use for an extract batch? For steeping specialty grains? These are all common questions.
Everyone has heard about brewing water adjustments, whether it’s an extract-based recipe calling for two tablespoons of gypsum or an all-grain recipe for Burton Ale that specifies mash water like that of Burton-upon-Trent. And, you may have read articles on how to calculate amounts of brewing salts to use, or why not to use them at all. Confused? Not sure when, why or how to adjust your water? Join the club, many other homebrewers aren’t sure, either.
How you treat and use your brewing water depends on your brewing method. Are you brewing with malt extract? Extract with steeped specialty grains? All-grain? When you are brewing from scratch with malted barley, water chemistry can affect every aspect of the wort, including fermentability, flavor and clarity. When you are brewing with malt extract, all of the mash-related decisions have been made for you. For extract brewing, the emphasis needs to be on flavor, starting with: does the water taste good? If the water tastes good, the beer should taste good. Any further adjustments, such as brewing salt additions, need to take a back seat to more important variables like sanitation, general water quality, pitching an adequate amount of yeast and fermentation temperature control.
General Water Quality
The first thing to do is to determine how your tap water smells and tastes. Is it sweet and refreshing, conjuring up images of snow-capped mountains? Or does it remind you of verdant green ponds or the YMCA swimming pool? Could you practically build a car out of it? If the water smells bad, many odors (including chlorine) can be removed by boiling. However, many city water supplies use a chemical called chloramine instead of chlorine to kill bacteria. Chloramine cannot be easily removed by boiling and will give a medicinal taste to your beer. But, chloramine can be removed by running the water through an activated-charcoal filter or by adding a Campden tablet (potassium metabisulfite). Charcoal filters are a good way to remove most odors and bad tastes due to dissolved gases and organic substances. These filters are relatively inexpensive and can be attached inline to the faucet or spigot. Campden tablets are another way to remove chloramine and should be readily available at your homebrew shop. One tablet will treat 20 gallons, so use half and crush it up to help it dissolve. Then proceed with the boil as usual. (If you use the whole tablet, it won’t hurt the beer flavor at all.)
Water softening systems can be used to remove bad-tasting minerals like iron, copper and manganese. Salt-based water softeners use ion exchange to replace these metals with sodium and potassium. Softened water works fine for extract brewing but it removes the calcium and magnesium, that are needed for all-grain beers. When you are brewing with softened water, be cautious when a recipe calls for adding gypsum to the wort. Water softeners do not effect sulfate levels in the water and the malt extract may already contain sulfate. The combination of high sulfate with the typically high sodium levels from water softening systems can produce a very harsh bitterness.
A good bet, if your tap water seems questionable, is the bottled water sold in most supermarkets. You should be able to get a mineral analysis of the bottled water by calling the manufacturer. I have done this with a couple of different brands. Bottled water is typically low in minerals like sulfate and carbonates, which makes it more adaptable for brewing.
Malt Extract Production
When you are brewing with malt extract, you are brewing with someone else’s dehydrated wort. When liquid malt extract is produced, the typical starting wort specific gravity is about 1.065. The wort is then concentrated by evaporation to 80% solids by weight. All of the minerals that were present in their water and in their mash are still in the malt extract.
It is common for extract producers to add gypsum to their lighter colored mashes to help achieve the proper mash pH in areas of moderately alkaline water. Therefore, before you add more gypsum to a hoppy pale ale recipe, make sure you brew it without any sulfate ion additions first, so that you can gauge the bittering potential of your extract as-is.
When you add water to brew an extract batch, you are adding still more minerals from your local water supply to those already present. So, you may be wondering if you should use distilled water to preserve the original mineral balance in the dehydrated wort. This isn’t really necessary, though it won’t hurt the situation. Most of the time, you can make an extract brew using your local water and you won’t be able to detect a negative difference in flavor over a batch made with distilled water. However, if you live in an area of very minerally, high hardness water, you may want to use some distilled or bottled water in your brewing.
Brewday Water Considerations
Questions about water do not end with pre-brewing water treatment. They extend to the actual water usage on brewday. How much water should you use for steeping? For boiling? Should I boil the water first and let it cool overnight? Should I chill the water in the refrigerator?
The only reason to boil your brewing water before brewday would be to drive off unpleasant odors, or to be sure of its sanitation if you are on a well. There is no need to boil it and decant it off the calcium carbonate that precipitates. That practice is for all-grain brewers trying to reduce the water’s alkalinity for mashing.
You have probably heard about the benefits of doing full-volume boils as opposed to partial boils containing all the malt extract. By doing a full volume boil, you are able to boil a lower gravity wort and prevent caramelization and darkening of the extract. In addition, a lower gravity boil will improve your hop utilization — you can get the same amount of bitterness out of fewer hops by boiling in a larger pot.
But what if you don’t have the firepower to boil the full five gallons? Well, you can get the same benefit by only boiling half of your extract with your hops, and steeping the rest. Here is what you would do:
1. Prepare a typical gravity wort (1.040–1.050) using about half your malt extract and your usual boiling pot.
2. Re-calculate your hopping schedule based on this wort gravity and volume.
3. Boil this small wort to achieve your desired hop bitterness, then turn off the heat and dissolve in your remaining extract. Most of the hot and cold break separation has occurred in the extract’s production, so the extract does not need to be boiled again.
4. Finally, cool this high-gravity wort and dilute it in your fermenter. Aerate and pitch your yeast as usual. You have achieved the benefits of a full volume boil without actually boiling the entire batch.
Steeping Specialty Grains
When you steep specialty grains, you aren’t trying to convert starches to sugars. You aren’t trying to maintain a particular temperature for a period of time; you are making tea. By placing crushed specialty grain in a mesh bag and soaking it in hot water, you are extracting color and flavors that are already there. If you leave the grain bag in the pot too long, or boil the bag, you can develop astringent tastes from the malt husks. This is most noticeable when steeping the darkest malts, like roast barley or black patent malt. You can reduce this tendency by a couple ways. One is to steep in a reduced volume of water, around a half gallon per pound of grain (4.2 L per kg). Another way is to use bicarbonate (either sodium bicarbonate or calcium carbonate) to balance the acidity of these dark malts. One teaspoon of sodium bicarbonate (baking soda) per 4 gallons (11 L) will add nearly 200 ppm of bicarbonate and will help you produce a smoother tasting stout.
Targeting a Beer Style
Many brewers are interested in duplicating a classic style from a famous brewing city. Depending on what your initial water chemistry is, you may want to add more sodium, chloride, sulfate, calcium or bicarbonate to better mimic a brewing city’s water. These ions can be added in the form of non-iodized table salt, sodium bicarbonate, calcium chloride or gypsum. This is a common desire for brewing Dortmunder Export, Burton Ale, etc., but let’s not put the cart before the horse. These styles grew out of the local water conditions, the brewers did not create the water to match their beer style. Water chemistry is hard to adjust because it is very interactive. The first time you try a salt addition to a recipe, you may want to cut the planned addition in half, because you don’t know how much of the ions the extract is contributing. It is better to undershoot an addition than overshoot and ruin the beer.
There is a big, dirty secret when it comes to matching the water of brewing cities— published water reports are usually annual averages, often from multiple sources and the numbers may not add up. This means that the listed profile may be physically impossible to achieve, that the combination of ion levels listed cannot exist together. Also, breweries commonly treat their water. There’s no guarantee a Dortmund brewery’s brewing liquor resembles untreated Dortmund water. So, you need to be satisfied with a result that is “close enough.” Flavor ion additions are added to the boiling pot where they are easier to dissolve. Gypsum does not dissolve well in plain water, even in hot plain water; it needs the acidity of the wort. Epsom salt (magnesium sulfate) may also be used, and is often a component of Burton Salt mixes you may see at a brewshop, but magnesium can be easily overdone resulting in off-flavors.
Brewing Water Adjustment
Now, before we begin, we all need to recite the First Rule For Adjusting Brewing Water: Know What Your Initial Water Chemistry Is.
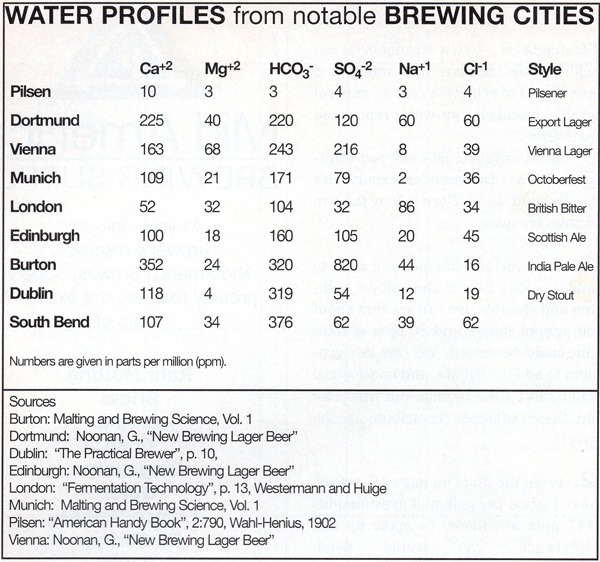
Attempting to adjust your brewing water without knowing what you have to begin with is like taking medicine without telling the doctor what’s bothering you. You have got to know what you are working with before you try fixing it. To calculate how much to add, use the table above to figure out what concentration is desired and then subtract your water’s concentration to determine the difference. Next, consult the table below to see how much of an ion a particular salt can be expected to add. The ion contributions for the salts are for 1 gram dissolved in 1 gallon (3.8 L) of distilled water. Unless you have a gram scale handy, you will probably want to convert the calculated weight of salt to volume, because it is more convenient. Teaspoon and milliliter equivalents for the salts are also given. Brewing software programs will let you calculate how much of a salt to add to achieve a certain mineral profile, based on your water report and batch size.

Summary
Water adjustment for extract brewing is a lot simpler than for all-grain brewing. When you brew with malt extract, all the questions of mash chemistry and wort fermentability have been made for you. Thirty years ago this was a problem because malt extract was primarily produced for baking – extract brewers and extract producers did not have the same goals. Malt extracts were commonly cut with corn syrup and other sugars. These days however, extract brewers and beer kit producers have the same goal — brewing excellent beer, so you only have to worry about tweaking the flavor to your personal taste.



