It’s Complicated: Understanding oxygen in brewing
Minimizing the formation and activity of ROS (O2-, HOO* , H2O2 and HO*) in beer and wort, must be a first step for improving beer flavour stability.” — Bart Vanderhaegen.

As brewers we get lots of conflicting information about the roles of oxygen in brewing. The reason there are so many opinions is that it is one of the most difficult and most important parameters to control. Until you have control over oxygen in your brewing process you won’t be able to make a high-quality beer. Oxygen is the primary parameter to control when concerned about shelf life.
Oxygen is a pesky molecule that can bond strongly to many compounds. While O2 is relatively stable, it is easily converted by iron and copper ions into much more reactive oxygen species (ROS), also known as free-radical oxygen. However, beer also contains antioxidants that will protect beer for the beginning of aging. Making this antioxidant effect outlast the expected shelf life is the goal of most beer styles. These antioxidants include melanoidins and reductones, or compounds that negate free-radical oxygen, from the malt. Melanoidins and reductones are more prevalent in darker malts and darker beers have a better shelf stability when compared to the lightest beers (this is true with roast malts, but crystal malts can lead to instability). Polyphenols from malt and hops will also bind to oxygen. While there are flavor changes associated with polyphenol reactions not all are considered detrimental. Interestingly, analysis of famous hazy IPAs shows both lots of polyphenols and lots of total package oxygen (TPO). The short shelf life of these beers suggests that lowering oxygen is more important than having lots of polyphenols.
Oxygen can also be bound enzymatically to linoleic acid from barley creating the famous wet paper staling flavor from trans-2-nonenal (T2N). While some new varieties of malt cannot produce this enzyme it is still important to eliminate oxygen ingress in brewing processes. For more on this topic, see my “Advanced Brewing” column titled “LOX-less Malts” in the January-February 2019 issue. Flavors that can be attributed to oxygen are Sherry, wet paper, cheesy (from improper hop storage), woody, ribes, black currant, catty, green apple, winey, and soapy.
It is convenient to think of different stages of the brewing process separately. We are going to go sequentially, first covering hot-side aeration, then onto post-chilling wort oxygenation, then the most critical to control: Post-fermentation oxygen pickup. After the yeast has metabolized the oxygen present at pitching all further oxygen sources should be eliminated. We say this is at the end of the lag period but the line is a little blurry.
Hot-Side Aeration
Nothing has gotten more interest in brewing in my career than hot-side aeration (HSA). When the first HSA paper came out linking staling flavors to aeration on the hot side of the brewery it was a shocking revelation. Oxygen can be introduced on the hot side in many places. The hot liquor can have dissolved oxygen. Milling the grain can entrain oxygen into the grist with the husks. Splashing during recirculation (vorlauf). Splashing into the grant. Exposure of the surface of the kettle during boil and whirlpool are all potential sources of hot side aeration.
With all of these possibilities and the difficulty of eliminating the sources of oxygen it begs the question: How important is HSA in my brewery? HSA has been linked to staling in packaged beer. There is no question about this. The question is how much and when will it occur. Even in the best of storage conditions the delicate flavors of a fresh beer are fragile. We can speed up a beer’s demise by storing it warm or cycling it between hot and cold and find out how our packaged beer might fair over time.
Oxygen is the primary parameter to control when concerned about shelf life.
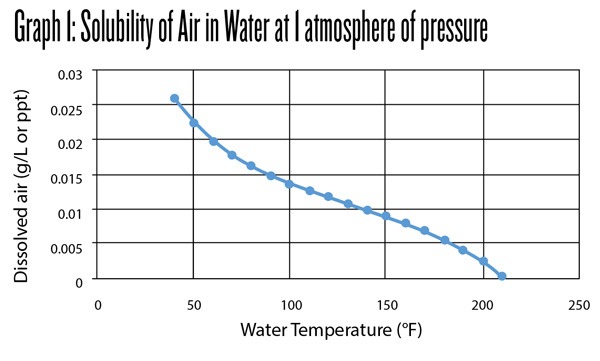
Obviously there are other gasses than oxygen in air, only about 21% of the atmosphere is comprised of oxygen. As you can see in Graph 1 below, as water is heated the gas is forced out. You will see this as you heat water in a pan. Bubbles of gas will start to form. However, we need to drive the gasses out of the water in order to remove them from the system or we end up with a supersaturated state. Boiling provides enough motion to remove dissolved gasses on our scale but a larger tank would need to provide other methods of agitation. While boiling will lower the O2 level of water, it will not eliminate the oxygen entirely and utilizing a dissolved oxygen (DO) meter is useful in finding the best process to remove O2.
The big debate in the beer world is: If my beer is going to be consumed within a month or two, how much concern do I need to pay to HSA? Charlie Bamforth and I have spoken at length about HSA. While his words are: Don’t worry about it as a small brewer. But when you ask him for specifics like, “can I just splash wort during recirculation?” His reply would be something along the lines of, well I wouldn’t. Sierra Nevada goes to great lengths to eliminate HSA. They do so by oxygen reduction, and minimizing any ions that might activate oxygen, in every step of their process. I find their product to be consistently high quality with a strong shelf life.

I follow a few homebrewing threads around the world and for some brewers going to the extremes of a pro brewer are appreciated. I have heard of brewers who claim that on day one their beers have a better malt presentation and are brighter than beers that have suffered HSA. In my brewing I do what I can to avoid HSA but I don’t go crazy. Chart 1 above provides some options. I have never heard of anyone, except lab workers, going to all of the extremes listed here. The one paper I have read that did go to all of the effort to completely eliminate HSA claimed that some of the necessary flavors were not created. Fortunately for us, we cannot ever eliminate all sources of HSA.
Wort Oxygenation
We go through all of this effort to eliminate air in all places except for one. After we cool the wort, we add oxygen as a yeast nutrient. This is extremely important if we don’t have healthy yeast. The oxygen allows a pathway that provides more energy to the cell and allows it to replenish depleted sterol reserves. It also prepares the cells to multiply, increasing our cell count. Big brewers traditionally use air to provide this oxygen as it is hard to over-oxygenate, but setting up sterile air is more difficult for the homebrewer than pure O2. But without an O2 meter you have to guess at the proper oxygenation level. A good test is to measure the amount of yeast added and make sure you recover 4x as much after fermentation. This is a pretty good indicator of proper oxygenation.
It is very important that we separate as much trub as possible before adding oxygen and lower the temperature to as close to pitching temperature as we can. Oxygenating trub can create off-flavors reported to be soapy and unpleasantly bitter. If we want to eliminate this step entirely it is possible if you multiply your yeast up to a fermenting cell count as opposed to a pitching cell count and make sure the yeast’s sterol reserves are maximized. Then you can eliminate the yeast oxygen step from the brewing process. Dry yeasts are typically dried at a point of very high sterol reserves and many brewers use them without adding wort O2.
Post-Fermentation Oxidation
On the cold side of brewing it is worth every effort to eliminate oxygen ingress. Even exposing the surface of beer to air is sufficient to pickup oxygen. In professional brewing where dissolved oxygen is often measured, levels are controlled in the parts per billion (ppb) range. Bottling lines that have less than 6 ppb DO pickup and canning lines that have less than 30 ppb DO pickup are commonplace. The only way to maintain the low levels of oxygen is to have strong quality control and quality assurance programs in place.
Before we move beer from one vessel to another it is important we make every effort to avoid splashing and purge the vessel of oxygen as much as possible. For transferring to a bucket we can only fill gently from the bottom with a hose. When transferring to a keg we can pressurize and vent a few times or if we have water to waste we can fill with water and purge with CO2.

One of the best ways to deal with oxygen is bottle/keg conditioning. This is the process of adding sugar and yeast to the final package and encouraging a final fermentation to carbonate the beer while scavenging oxygen. In normal homebrewing there is enough yeast in the beer at the end of fermentation to eliminate the yeast addition and one only needs to add sugar.
Last week I was cleaning up and had two special bottles of beer. They were 100% Maris Otter and wet hopped with Centennial that I bottled 260 Champagne bottles of in 2006. Because the tank had been clarified, I added dried yeast and sugar individually to each bottle before filling it. The bottles were sealed with a Champagne cork and hand numbered and two extra bottles were numbered 0/260 and 261/260. I saved the two extras. Thirteen years later I had no hope for the beer having survived but I also no longer had room for sentimental bottles so I opened them.
Surprisingly, the beers were delicious. The malt had become more toffee-like, there was no hop character at all and a phenolic-producing yeast had made a very slight clove character. Neither the pitching nor the bottling yeast have the gene to produce phenolics so this was a contamination. The beer was crystal clear and golden. The dry fermentation and the sweetness from aging balanced well to make a very refreshing and drinkable beer. We will call those lucky bottles and I would not expect to be able to repeat these results.
I have had many private conversations with Quality Assurance personnel at many breweries and I have included their advice without attribution to protect the proprietary processes of the breweries involved. It has helped me to refine my beers and hopefully it can help you as well.
References:
Wort Trub Content and Its Effects on Fermentation and Beer Flavor – D. O. Schisler, J. J. Ruocco, and M. S. Mabee, Adolph Coors Company, Golden, CO 80401
“The chemistry of beer aging – a critical review” – Bart Vanderhaegen, Hedwig Neven Hubert Verachtert, Guy Derdelinckx
During the technical review process tangential conversations can break off between reviewer and author that can be interesting and may at times become folded into the story. We thought this back and forth between “Advanced Brewing” columnist Colin Kaminski and Technical Editor Ashton Lewis was worthy of its own space.
Ashton Lewis: Graph 1 is a great depiction, but it includes an assumption that I cannot prove or disprove with references . . . and I have searched for a long time in the past. The assumption is that boiling water contains no oxygen. The problem with this argument is that the boiling water is still exposed to atmospheric pressure and the absolute pressure is not zero. If I understand Henry’s Law, the system is still under pressure and gas solubility should be more than zero. Water deaeration in a commercial brewery is more extreme than simply heating to 212 °F/100 °C.
Colin Kaminski: While visiting a highly successful 15-bbl brewery and talking water, they had a scaled down copy of an expensive commercial deaeration system. The brewer had been using an old boil kettle to boil and cool water before the system. After months of work, the kettle was still better at lowering the O2 level for hot liquor than the deaeration system, but you are right, neither could get to 0. The final data point in Graph 1 is dissolved air/water ratio at 210 °F (99 °C) = 0.0004 g/L.
Ashton Lewis: 0.0004 g/L (ppt) x 1,000,000 x 35% O2 = 140 ppb. (35% is the approximate calculated from Henry’s Law level of O2 dissolved from a nitrogen, O2, CO2 blend matching the atmosphere.) This is a very low value but is not zero. If beer with 0 ppb of oxygen is diluted with water at a ratio of 1 part water to 4 parts beer, the resultant concentration is 28 ppb. This may be considered low for packaged beer, but it’s not very low for beer out of a fermenter. Interesting stuff!
Colin Kaminski: This is important for anyone looking to add water to beer post-fermentation. It is hard to see on Graph 1 but heating the water to 210 °F (99 °C) will not completely eliminate the O2 and this water would not be suitable for dilution of finished beer.



