Crystallization: Forming the ice in your eisbock, baby
Eisbock is a style of beer that is characterized by a strong, rich, warming aroma and flavor profile. Eisbock beers are produced by allowing an already-strong doppelbock to partially freeze. (See the October 2012 issue of BYO for how to brew a doppelbock.) The ice crystals that form are then removed and the liquid component that remains contains the concentrated alcohol and flavor compounds that were originally in the entire volume of beer prior to freezing.
To make a good eisbock (or any other kind of ice beer) it is helpful to have a good understanding of how ice crystals form, and to be able to effectively manage crystal formation within the beer.
Crystal formation
Ice crystal formation within a beer happens as a result of cooling the beer down to the freezing point of the solution or below. As the temperature is lowered, the vibrational energy of the water molecules becomes lower and lower until the point is reached where the vibrational energy is unable to overcome the intermolecular attractive forces between the water molecules. The individual water molecules join together and form an orderly, bonded structure with each other — ice (see Figure 1, below).
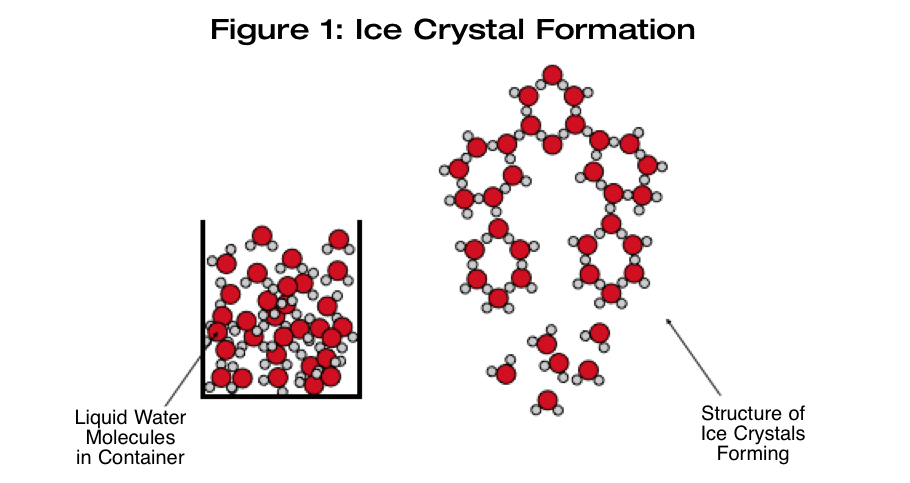
There are two steps involved in the formation of solid crystals within a liquid. The molecules must first form a solid particle in the liquid. This initial solid particle formation is called nucleation. This solid particle must then increase in size within the liquid. This step is (unsurprisingly) called growth.
The growth process involves two steps. First, diffusion of molecules within the liquid to the surface of the crystal solid/liquid interface must happen. The molecules that are at the interface must then be incorporated into the lattice of the crystal. Ice crystals form hexagonal plates that grow both by becoming larger hexagons and by forming a column. One interesting property of ice is that it expands and becomes less dense as it makes the transition from liquid water to solid ice. This is why ice floats. It is also why you should avoid making ice beer in a glass carboy. If the surface of the beer freezes solid, the pressure exerted by the expanding ice can easily crack the glass.
Crystal nucleation and crystal growth rate
Nucleation and growth rate are two defining parameters within the overall crystallization process. Figure 2 (below) shows a generalized time and temperature relationship for the crystallization process as it occurs within a pure water system, and provides a description for what is happening at specific points within the process.
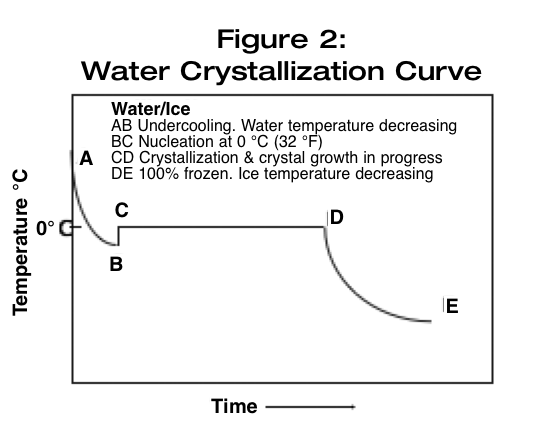
Initially, the temperature of water decreases and even temporarily dips below the freezing point, while still remaining liquid. Once the crystallization process begins, the temperature of the system remains at the freezing point while water is frozen into ice. (The system is losing heat over time to the crystallization process, but the temperature does not change.) Once the water is frozen, the temperature of the ice will begin decreasing.
Nucleation
Nucleation is the word used to label the initial formation of solid particles within liquid at the molecular level. It is to this “core” of solid particles the other molecules then attach and form the crystal. The molecules forming a crystal nucleus must resist the tendency to return to the liquid state, and also must become oriented into a fixed lattice.
The mechanism of crystal nucleation from solution has been studied by many scientists, and their work suggests that the nucleation rate is the sum of contributions by (1) primary nucleation and (2) nucleation due to contact between the crystals, the walls of the container, or the impeller mixing the system. This is stated succinctly as:
B0 = Bss + Bcc + Bci
Where:
B0 = the net number of new crystals formed in a unit volume of liquid per unit of time
Bss = the primary nucleation rate due to the thermodynamic driving force
Bcc = the rate of nucleation due to crystal-crystal and crystal-container contacts
Bci = the rate of nucleation due to crystal-impeller contacts
Impellers are used in laboratory situations to keep the temperature of the liquid uniform. Obviously, in a homebrew setting, it is highly unlikely you will be mechanically stirring your eisbock. If an impeller is not used, the last term in the equation becomes zero. The probability that a stable crystal nucleus will form depends on many factors including activation energies, lattice bond energies and the presence of impurities (which affect overall system energies). The minimum number of water molecules required to form a stable crystal nucleus has been estimated to be between 80-100.
Crystal growth rate
Crystal growth is a layer-by-layer process. Crystal growth can only occur at the face of the crystal. Water molecules must be transported to the crystal face from the bulk of the liquid via diffusion. Diffusional resistance to the movement of molecules to the growing crystal face, as well as the resistance to integration of those molecules into the face, must be considered.
If L is defined as a characteristic dimension of the crystal, the rate of growth of a crystal face that is perpendicular to L is defined as:

Where:
G = the growth rate over time interval t
ΔL = change in the length of characteristic dimension of the crystal
Δt = time interval
Crystal growth rate, when defined in this way, is independent of crystal size, provided that all crystals in the suspension are treated alike.
A mathematical model has also been developed which can correlate the nucleation rate to the crystal growth rate. Because the crystal growth rate is more easily determined than nucleation rate, and because nucleation is a very nonlinear phenomena, a model of this form is used:
B0 = ksb
Where:
B0 = the net number of new crystals formed in a unit volume of liquid per unit of time
s = crystal saturation extent (mass of total system – mass of crystals present)
b = experimentally derived constant
k = dimensional coefficient specific to the material
The form of this model implies that the more crystals that are already present in the system, the slower the formation of new primary nucleation sites will be. This makes intuitive sense, as it becomes easier and easier for a water molecule to find a crystal and attach as the number of already-existing crystals within the system increases. It is generally more thermodynamically favorable for a water molecule to attach to an existing crystal than to begin to form a new nucleation site.
Practical considerations for brewing
Crystal size strongly depends upon the rate of freezing. A fast freezing rate promotes the formation of many small ice crystals. A slower freezing rate promotes growth of larger crystals. If the beer is cooled quickly, the freezing will happen faster, but the ice crystals that are formed will be smaller, on average, than crystals in beer that was frozen more slowly. Smaller ice crystals may be more difficult to remove from the beer than larger crystals.

Agitation increases primary nucleation rate and therefore crystallization rate within the system. If faster freezing with correspondingly smaller crystals is desired, shake the container of beer when the ice crystals initially begin to form in order to increase the number of “nucleate” crystals. If you desire larger, more easily-removed crystals, avoid agitation of the container of beer during the freezing process.
If your beer freezes solid, it can exert a tremendous amount of pressure on the walls of the vessel. Strong beers such as doppelbocks tend to form slushy mixtures and may take a very long time to freeze solid in a freezer. Still, it is prudent to check on them periodically, just in case.



